Quality Control: Enhancing Accuracy and Reliability
In response to the critical need for medical laboratory personnel to comprehend the significance of Quality Control (QC) strategies and propose high-quality practices for reinforcing accuracy and reliability in diagnostic testing, this article embarks on unraveling the complexities of QC in clinical laboratories. We delve into the vital components and approaches of QC, aiming to equip laboratory professionals with the understanding and tools essential to uphold stringent standards of accuracy and reliability in diagnostic testing. By exploring the nuances of QC practices and aligning with regulatory pointers, this article seeks to empower clinical laboratory personnel.
Understanding Quality Control in Clinical Laboratories:
Quality control in clinical diagnostics is vital for ensuring precise and reliable test outcomes that are essential for patient management. It involves meticulously monitoring procedures to maintain accuracy and consistency in laboratory testing, contributing to high-quality patient care.
Essential Components of Quality Control:
An effective QC program comprises:
- internal quality control (IQC)
- external quality assessment (EQA)
- proficiency testing
- calibration
- staff training.
These components collectively uphold the accuracy and precision of laboratory results by ensuring standardized practices and meticulous oversight.
Internal Quality Control (IQC):
Internal quality control involves monitoring test performance within the laboratory using known control materials. By running internal control samples alongside patient samples, laboratories can assess the accuracy and precision of test results. For example, in a biochemistry lab measuring glucose levels, running daily control samples at different concentration levels ensures the accuracy of the glucose assay.
The IQC process involves systematically monitoring and evaluating test performance using known control materials to ensure the reliability of testing procedures. To execute the IQC process effectively, laboratories typically follow these steps:
-
- Selecting Control Materials: Choose control samples with known values that represent the expected range of test results to be monitored.
- Running Control Samples: Incorporate control samples along with patient samples during testing procedures to assess the accuracy and precision of test results.
- Analyzing Results: Compare the measured values of control samples to their expected values, identifying any discrepancies or trends that may indicate variations in test performance.
- Taking Corrective Actions: If deviations are detected, investigate the root cause, implement corrective actions, and document the steps taken to maintain the quality and reliability of test results. By diligently executing internal quality control procedures, laboratories can identify and rectify errors promptly, ensuring the accuracy and consistency of diagnostic testing and ultimately enhancing patient care and outcomes.
External Quality Assessment (EQA):
External quality assessment involves laboratories participating in external proficiency testing programs to benchmark their performance against peer laboratories. For instance, a microbiology lab analyzing bacterial cultures may participate in an EQA program where identical samples are distributed to multiple labs for analysis. Discrepancies in lab results highlight areas for improvement in testing procedures.
The EQA process involves participating in proficiency testing programs where laboratories receive blind samples to analyze alongside peer laboratories. To effectively execute the EQA process, laboratories follow these key steps:
-
- Enrollment in Proficiency Programs: Join EQA programs specific to the types of tests performed in the laboratory to assess proficiency in testing procedures.
- Analyzing Blind Samples: Receive and analyze EQA samples without prior knowledge of their values, comparing results with expected outcomes provided by the EQA program.
- Reviewing Performance Reports: Assess performance reports from the EQA program, identifying any discrepancies or trends in results compared to peer laboratories.
- Implementing Corrective Actions: Address any identified issues through corrective actions, including reevaluation of testing procedures, staff training, or equipment maintenance to enhance testing accuracy and reliability. By actively participating in external quality control programs and consistently evaluating and improving testing performance based on EQA results, laboratories can enhance the overall quality of diagnostic testing, ensuring precise and reliable test outcomes for optimal patient care.
Proficiency Testing:
Proficiency testing evaluates the competence of laboratory staff in performing specific tests accurately and reliably. In a hematology lab, proficiency testing for a complete blood count (CBC) may involve analyzing samples with known parameters and comparing the results with expected values. Regular proficiency testing ensures staff competency and test result validity.
The proficiency testing process involves laboratories receiving samples with known parameters to analyze and compare their results with expected values. To successfully execute proficiency testing in the lab, laboratories typically follow these steps:
-
- Enrollment in Proficiency Programs: Register for proficiency testing programs relevant to the tests conducted in the laboratory to assess staff competency and testing accuracy.
- Analyzing Proficiency Samples: Receive and analyze proficiency testing samples with known values, ensuring testing procedures are performed according to established protocols.
- Comparing Results: To evaluate the accuracy and reliability of test outcomes, compare the laboratory’s results with the expected values provided by the proficiency testing program.
- Implementing Corrective Actions: Address any discrepancies or deviations identified during proficiency testing by investigating the root cause, taking corrective actions, and documenting the steps to improve testing procedures. By actively engaging in proficiency testing and using the results to enhance the skills and performance of laboratory staff, laboratories can uphold standards of accuracy and reliability in diagnostic testing, ultimately contributing to high-quality patient care and diagnostic outcomes.
Calibration:
Calibration is the process of adjusting and standardizing laboratory instruments to ensure accurate and consistent measurements. In a molecular diagnostics lab performing polymerase chain reaction (PCR) tests, calibrating the thermal cycler using known DNA concentrations guarantees the reliability of subsequent test results. Regular equipment calibration is essential for maintaining measurement accuracy.
Calibration protocols within the laboratory are important for ensuring the accuracy and reliability of test results by standardizing and adjusting equipment to recognized reference standards. The calibration manner involves comparing the measurement produced by the instrument to a known standard or reference to detect and correct any deviations. Calibration protocols typically include the following steps:
-
- Establishing Reference Standards: Identify and procure reference materials or standards representing known values for the measured parameters.
- Setting Calibration Intervals: Determine how often calibration is required based on factors like instrument stability, usage frequency, and manufacturer recommendations.
- Performing Calibration Procedures: Follow manufacturer guidelines to adjust instrument settings, calibrate sensors, and align measurements with reference standards.
- Recording Calibration Data: Document calibration dates, results, adjustments made, and any deviations from reference standards for traceability and quality assurance purposes. Effective calibration ensures that laboratory instruments provide accurate and consistent measurements, ultimately contributing to the reliability of test results and high-quality diagnostic outcomes.
Staff Training:
Staff training is crucial for ensuring that laboratory personnel are competent in performing testing procedures accurately and following established protocols. In a histopathology lab, training on tissue processing techniques ensures uniform and consistent sample preparation for microscopic analysis. Continuous training and competency assessments are essential for upholding testing standards.
The essential components of quality control—internal quality control, external quality assessment, proficiency testing, calibration, and staff training—are integral to maintaining the accuracy and reliability of diagnostic testing in clinical laboratories. By implementing these components effectively and utilizing practical examples, laboratories can uphold high testing quality standards, ensuring precision in patient care and diagnostic outcomes.
By practicing quality control procedures, clinical laboratories can enhance the reliability and accuracy of their diagnostic testing, ultimately contributing to improved patient care and outcomes.
Creating a QC Plan
Creating a QC plan requires laboratories to integrate these components into a structured framework that ensures the accuracy and reliability of test results. Following this, compliance with regulatory standards such as those set forth by the Clinical Laboratory Improvement Amendments (CLIA) becomes paramount.
CLIA provides recommendations and guidelines for establishing quality control protocols, including adopting Individualized Quality Control Plans (IQCPs) tailored to specific test systems and environments. By incorporating the principles of IQCPs and adhering to CLIA recommendations, laboratories can enhance their quality control practices, uphold regulatory compliance, and ultimately improve the accuracy and reliability of diagnostic testing processes.
Introducing CLIA’s Step-by-Step Guide – IQCP Individualized Quality Control Plan:
The document provides a comprehensive guide to developing an Individualized Quality Control Plan (IQCP) in clinical laboratories as mandated by CLIA regulations. This approach allows labs to tailor QC procedures to their unique environments and patients, enhancing flexibility and regulatory compliance. The IQCP process includes Risk Assessment, Quality Control Plan (QCP) development, and Quality Assessment (QA) steps to ensure precise and reliable test results.
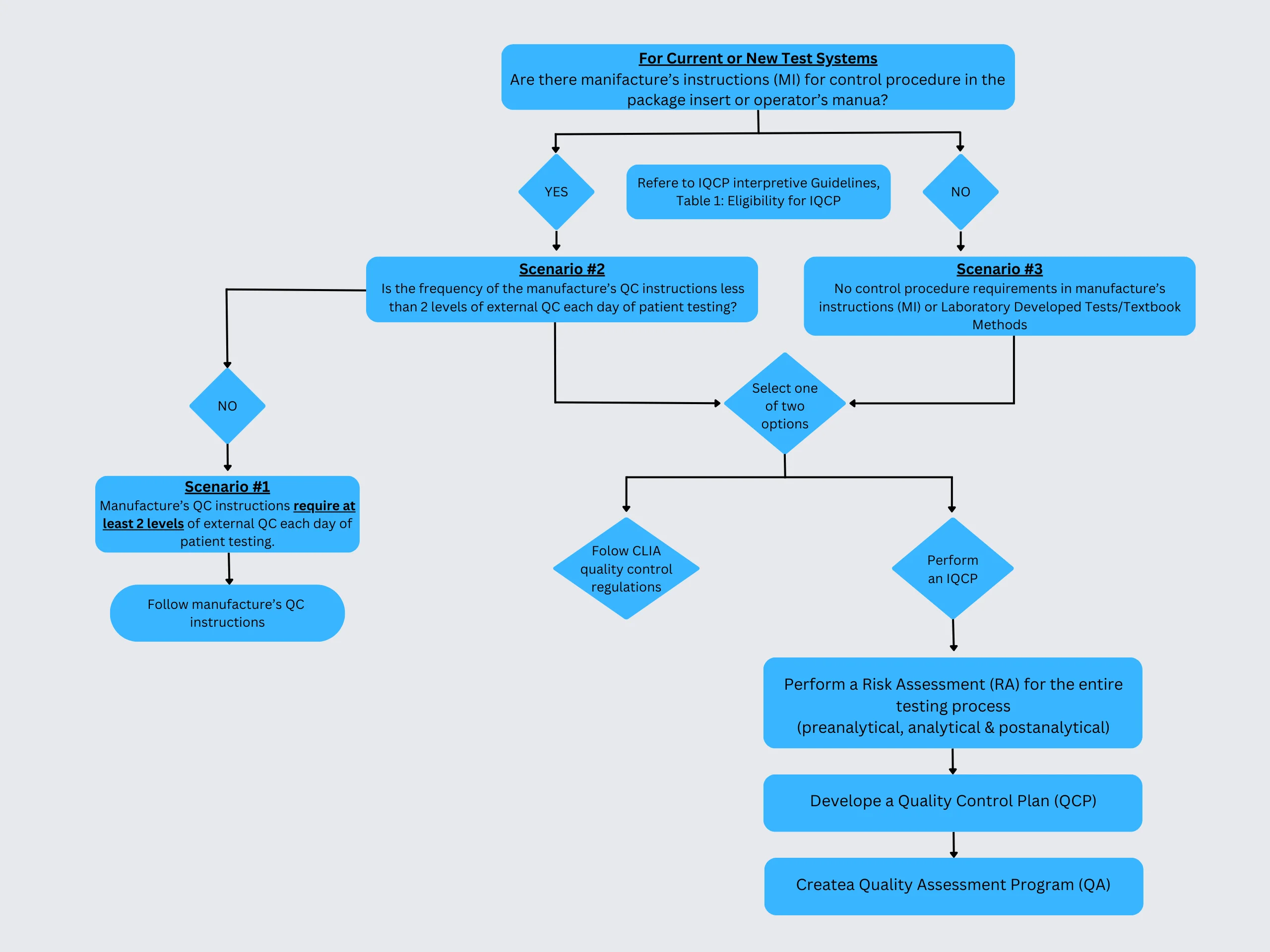
Discussion on CLIA’s Instructions for QC:
CLIA regulations mandate laboratories to implement QC procedures to monitor testing accuracy. The IQCP option enables labs to customize QC plans for specific test systems, integrating individuality and precision into the QC process. Adopting an IQCP allows labs to design tailored QC procedures based on various factors, enhancing the overall quality control framework.
Step-by-Step Instruction List for QC Labs:
Risk Assessment:
-
- Identify potential failures and sources of errors in the testing process.
- Evaluate specimens, test systems, reagents, environment, and personnel risks.
Quality Control Plan (QCP):
-
- Develop a comprehensive QCP outlining procedures to enhance accuracy and reliability in test processes.
- Include electronic controls, proficiency testing, calibration, maintenance, and staff training in the QCP.
Quality Assessment (QA):
-
- Continuously monitor the effectiveness of the QCP.
- Document errors, ensure staff qualifications, conduct training, and implement corrective actions as necessary.
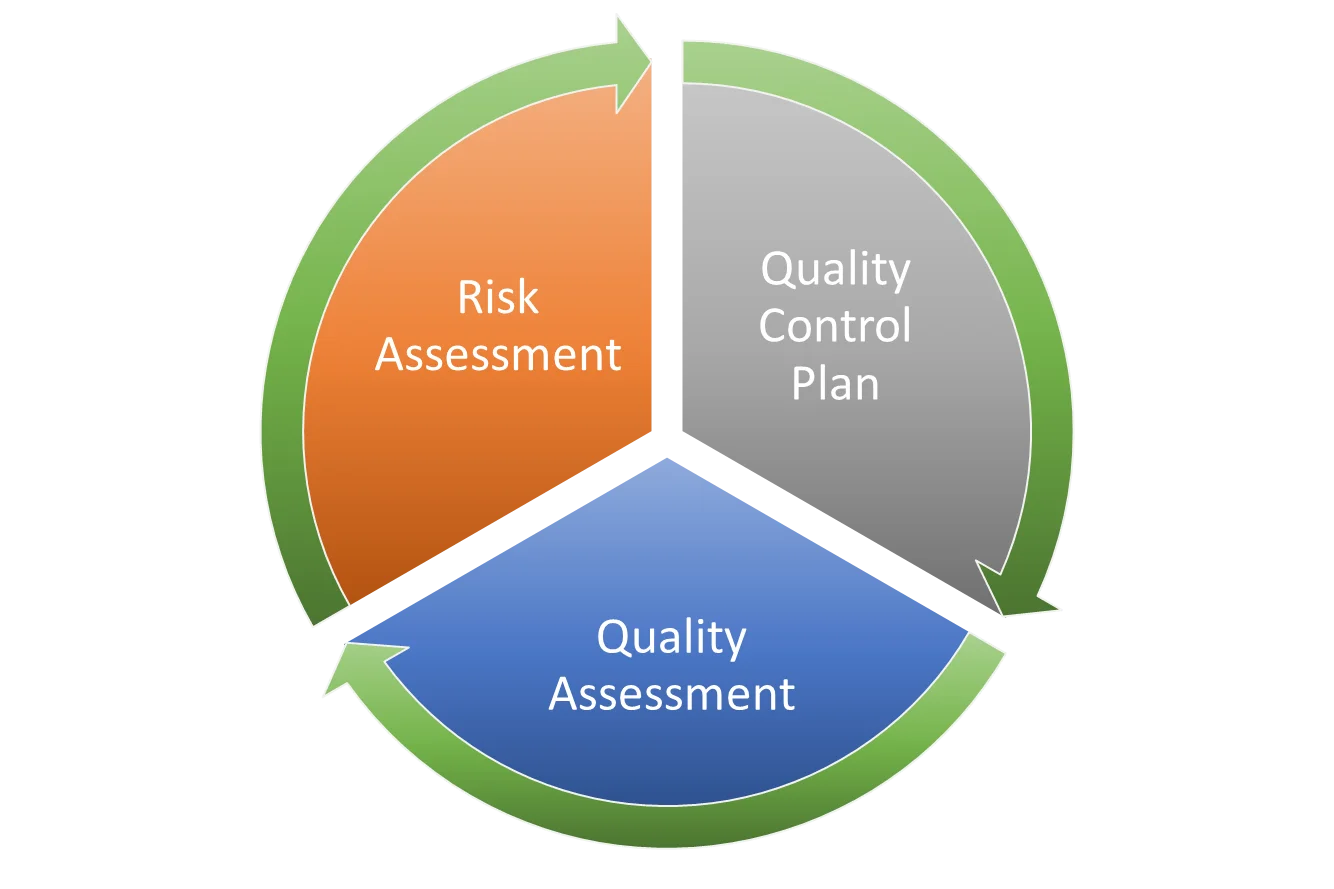
By following these step-by-step instructions, laboratories can establish a robust quality control framework compliant with CLIA regulations, promoting accuracy, reliability, and quality in diagnostic testing processes, ultimately advancing patient care and regulatory compliance.
Sources:
IQCP INDIVIDUALIZED QUALITY CONTROL PLAN: Developing an IQCP; A Step-By-Step Guide.



